智通财经APP获悉,10月13日,CDE官网显示,艾伯维(ABBV.US)注射用E型肉毒毒素(TrenibotE)上市申请获受理,注册分类为1类。根据公开资料和临床试验进展,Insight数据库推测适应症为中度至重度眉间纹。
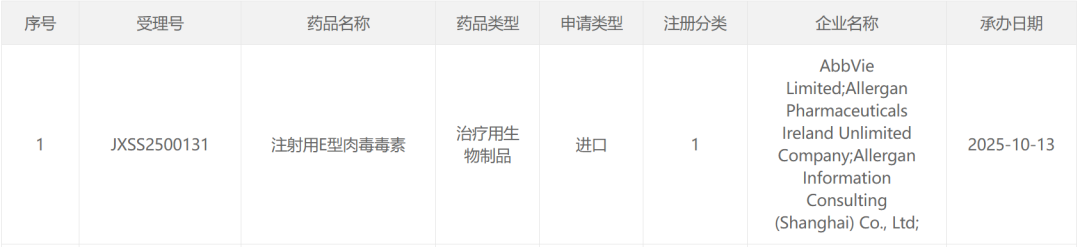
截图来源:CDE官网
TrenibotE是同类首创的E型肉毒神经毒素,2025年4月,艾伯维向FDA提交了该产品的首个上市申请,此申请得到来自2100多名接受TrenibotE治疗患者的临床研究数据的支持,其中包括两项评估TrenibotE治疗中度至重度眉间纹的关键性III期临床研究(M21-500和M21-508)以及一项III期开放标签安全性研究(M21-509)。
III期研究的所有主要和次要终点均已达到,给药后8小时即可快速起效(最早评估时间),且疗效持续时间为2-3周。此外,无论是作为单次治疗还是最多连续三次治疗,TrenibotE的治疗中出现的不良事件与安慰剂相似。
Disclaimer: Investing carries risk. This is not financial advice. The above content should not be regarded as an offer, recommendation, or solicitation on acquiring or disposing of any financial products, any associated discussions, comments, or posts by author or other users should not be considered as such either. It is solely for general information purpose only, which does not consider your own investment objectives, financial situations or needs. TTM assumes no responsibility or warranty for the accuracy and completeness of the information, investors should do their own research and may seek professional advice before investing.
